





























 Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.  Limited Edition Golden Llama is here! Check out how you can get one.
Limited Edition Golden Llama is here! Check out how you can get one.
 Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!  Offering SPR-BLI Services - Proteins provided for free!
Offering SPR-BLI Services - Proteins provided for free!
 Thank you for choosing ACROBiosystems. Would you rate our product and service?
Thank you for choosing ACROBiosystems. Would you rate our product and service?  Thank you for choosing ACROBiosystems. Would you rate our product and service?
Thank you for choosing ACROBiosystems. Would you rate our product and service?
 Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!  Here come GMP Grade Cytokines!Free Sample is available!
Here come GMP Grade Cytokines!Free Sample is available!
Human
Purified Human CD3ε Protein.
APC
Excitation Wavelength: 640 nm
Emission Wavelength: 661 nm
The Isotype control is sold separately and you can search for Cat. No. DNP-AFM487 for product information.
Lyophilized from 0.22 μm filtered solution in PBS, 0.03% Proclin 300, pH7.4 with trehalose as protectant.
Contact us for customized product form or formulation.
Please see Certificate of Analysis for specific instructions.
For best performance, we strongly recommend you to follow the reconstitution protocol provided in the CoA.
For long term storage, the product should be stored at lyophilized state at -20°C or lower.
Please protect from light and avoid repeated freeze-thaw cycles.
This product is stable after storage at:
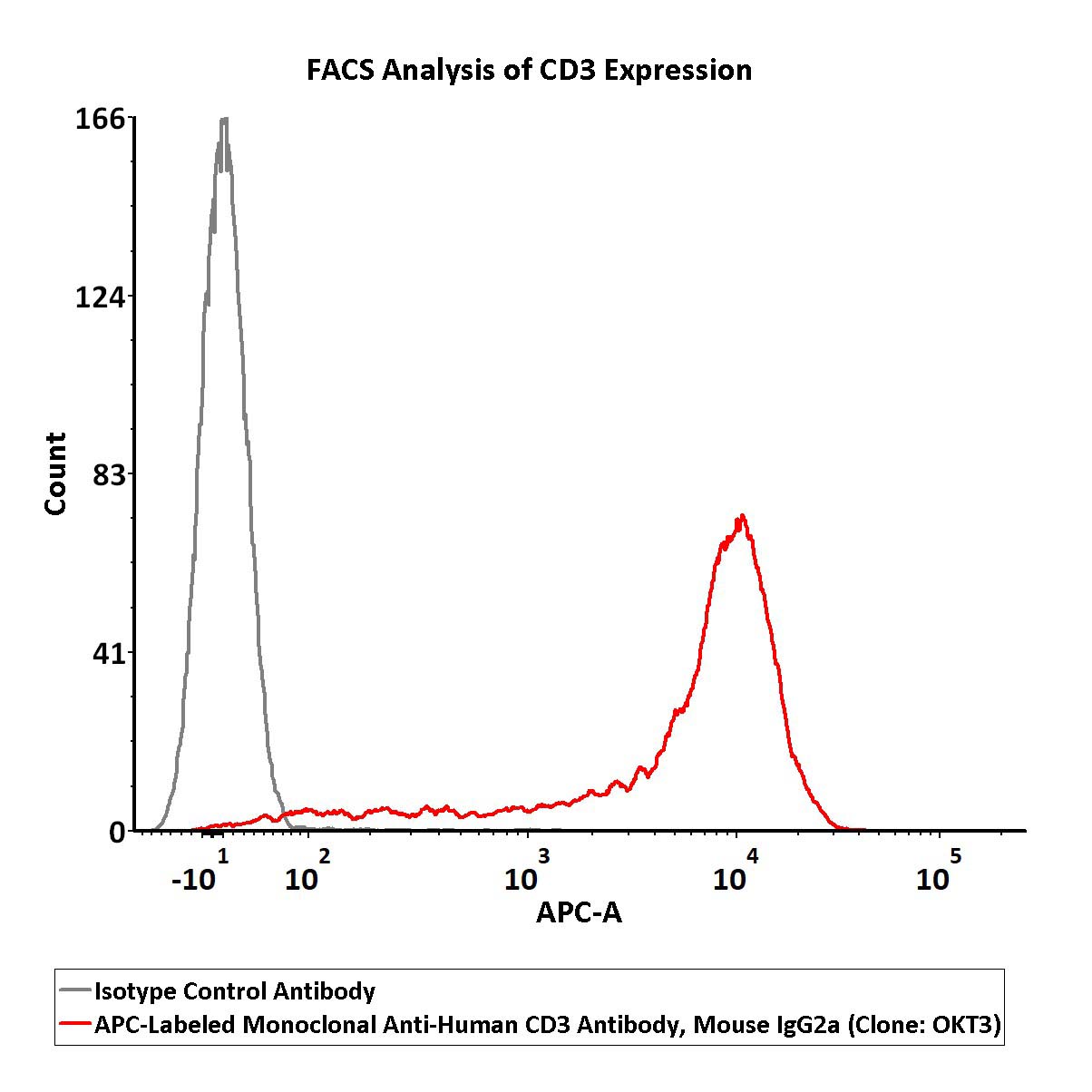
Flow cytometric analysis of Jurkat cells staining with APC-Labeled Monoclonal Anti-Human CD3 Antibody, Mouse IgG2a (Clone: OKT3) (Cat. No. CDE-AHFP1) at 1:50 dilution (2μL of the antibody stock solution corresponds to labeling of 1e6 cells in a final volume of 100 µL) , compared with isotype control antibody. APC signal was used to evaluate the binding activity (QC tested).

ACROBiosystemsは、二重特異性抗体の治療薬への臨床開発を加速するために、高い生物活性を持つ均一なCD3δ/CD3εおよびCD3γ/CD3εタンパク質のシリーズを開発しました。

We offer a wide range of cell and gene therapy solutions starting from discovery to the clinic. Explore our wide range of proteins, antibodies, kits, and other assays to accelerate the development of your cell and gene therapy.

Organoid Toolbox は、即使用可能のオルガノイド、オルガノイド分化ツールキット、創薬プロジェクトの進行を加速する、さまざまなサービスを含むオルガノイドソリューションのことです。

IL-15、IL-7、IL-21など、高品質のGMP準拠サイトカイン製品を提供しており、免疫細胞治療薬の臨床研究をサポートし、医薬品規制当局の承認を加速できます。

注目されている50以上のCAR-Tターゲットがあり、CARの発現の検出のために設計された抗原タンパク質をフローサイトメトリーで検証し、CARの発現が高い特異性で検出できます。ロット間の一貫性が保証されています。また、PE / FITC標識タンパク質を利用することで、ワンステップ染色でCARの発現を高い特異性でバックグラウンドなしに検出できます。

CD20、Claudin18.2、CD133、GPRC5D、CCR5、CCR8などの安定で高活性な全長型膜貫通タンパク質は、免疫学、ELISA、SPR、BLI、細胞実験、CAR陽性率検出に利用できます。VLP、界面活性剤、ナノディスクなどのテクニカルプラットフォームによって複数回膜貫通タンパク質の医薬品創薬を促進できます。

GMP準拠サイトカイン、高品質の細胞活性化/増幅試薬、遺伝子改変試薬/酵素、CAR検出試薬などの製品を提供し、細胞、遺伝子治療用に全体的なソリューションを提供し、創薬から臨床研究までのすべての段階をサポートします。

これまでに知られている免疫チェックポイント分子をほぼ製品化しており、様々なタグの製品群を提供できます。天然高分子の構造はMALSによって、生物活性はELISA/SPR/BLI/FACSなどによって検証済みです。またBiotin/FITCなどの標識も選択でき、抗体のハイスループットスクリーニングに利用できます。

弊社ACROBiosystemsはADC医薬品の開発のサポートに取り組み、注目されている様々なターゲットのために、異なる種類とタグの製品が開発され、高い純度と親和性が特長です。免疫、抗体スクリーン、SPR、細胞活性検査などの実験に利用できます。Protocolも無償で提供いたします。

すべての分子のFc受容体タンパク質だけでなく、一般的な変異体やビオチン標識タイプも含まれております。お客様のモノクローナル抗体の開発をサポートします。

インターロイキン、成長因子、ケモカイン、TNF など、様々なサイトカインターゲットの自然なコンフォメーションを確保するために HEK293 によって発現されます。SDS-PAGE/HPLC/SEC-MALS によって高純度は確認され、高い生物活性は ELISA/SPR/BLI によって確認されています。

AneuroはACROBiosystemsが神経科学研究のために設計した製品群のブランドです。神経科学研究を推進するための治療・診断用研究タンパク質、PFF、組み換え神経因子など、高品質の重要なタンパク質を提供しております。
| English Name | Research Code | Research Phase | Company | First Brand Name | First Approved Country | First Indication | First Approved Company | First Approved Date | Indications | Clinical Trials |
|---|---|---|---|---|---|---|---|---|---|---|
| Mouse monoclonal antibody against human CD3 antigen of T lymphocyte (Wuhan Institute of Biological Products) | Approved | Wuhan Institute Of Biological Products Co Ltd | Mainland China | Rejection of organ transplantation | Wuhan Institute Of Biological Products Co Ltd | 1999-01-01 | Rejection of organ transplantation | Details | ||
| Tebentafusp | ImmTAC-gp-100; IMC-gp-100; IMC-gp100 | Approved | Immunocore Ltd | KIMMTRAK | United States | Uveal melanoma | Immunocore Ltd | 2022-01-25 | Uveal melanoma; Melanoma | Details |
| Teclistamab | JNJ-64007957; JNJ-64007959; JNJ-7957 | Approved | Johnson & Johnson Innovative Medicine, Genmab A/S | TECVAYLI, TECAYLI | EU | Multiple Myeloma | Janssen-Cilag International Nv | 2022-08-23 | Hematologic Diseases; Hematologic Neoplasms; Multiple Myeloma | Details |
| Teplizumab | MGA-031; PRV-031; hOKT3-γ1-ala-ala; hOKT3-gamma-1-ala-ala | Approved | Tolerance Therapeutics Inc | TZIELD | United States | Diabetes Mellitus, Type 1 | Provention Bio Inc | 2022-11-17 | Diabetes Mellitus, Type 1; Rejection of renal transplantation; Glucose Intolerance; Hypoglycemia; Psoriasis; Diabetes Mellitus, Experimental | Details |
| Mosunetuzumab | BTCT-4465A; RO-7030816; CD20-TBD; RG-7828 | Approved | Genentech Inc | Lunsumio | EU | Lymphoma, Follicular | Roche Registration Gmbh | 2022-06-03 | Lymphoma, B-Cell; Lymphoma, B-Cell, Marginal Zone; Lymphoma, Large B-Cell, Diffuse; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Follicular; Lupus Erythematosus, Systemic; Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Epcoritamab | GEN-3013; ABBV-GMAB-3013 | Approved | Genmab A/S, Abbvie Inc | Tepkinly, TEPKINLY, EPKINLY | United States | Lymphoma, Large B-Cell, Diffuse | Genmab Us Inc | 2023-05-19 | Lymphoma, B-Cell; Lymphoma, B-Cell, Marginal Zone; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Follicular; Lymphoma, Mantle-Cell; Primary mediastinal B cell lymphoma; Richter's Syndrome; Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Blinatumomab | BiTE-MT-103; bscCD19xCD3; AMG-103; MT-103; MEDI-538 | Approved | Micromet Inc | Blincyto, 倍利妥 | United States | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Amgen Inc | 2014-12-03 | Leukemia; Leukemia, Myelogenous, Chronic; Neoplasm, Residual; Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Multiple Myeloma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor T-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Philadelphia Chromosome; Lymphoma, Non-Hodgkin; Burkitt Lymphoma; Lymphoma; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Mouse monoclonal antibody against human CD3 antigen of T lymphocyte (Wuhan Institute of Biological Products) | Approved | Wuhan Institute Of Biological Products Co Ltd | Mainland China | Rejection of organ transplantation | Wuhan Institute Of Biological Products Co Ltd | 1999-01-01 | Rejection of organ transplantation | Details | ||
| Tebentafusp | ImmTAC-gp-100; IMC-gp-100; IMC-gp100 | Approved | Immunocore Ltd | KIMMTRAK | United States | Uveal melanoma | Immunocore Ltd | 2022-01-25 | Uveal melanoma; Melanoma | Details |
| Teclistamab | JNJ-64007957; JNJ-64007959; JNJ-7957 | Approved | Johnson & Johnson Innovative Medicine, Genmab A/S | TECVAYLI, TECAYLI | EU | Multiple Myeloma | Janssen-Cilag International Nv | 2022-08-23 | Hematologic Diseases; Hematologic Neoplasms; Multiple Myeloma | Details |
| Teplizumab | MGA-031; PRV-031; hOKT3-γ1-ala-ala; hOKT3-gamma-1-ala-ala | Approved | Tolerance Therapeutics Inc | TZIELD | United States | Diabetes Mellitus, Type 1 | Provention Bio Inc | 2022-11-17 | Diabetes Mellitus, Type 1; Rejection of renal transplantation; Glucose Intolerance; Hypoglycemia; Psoriasis; Diabetes Mellitus, Experimental | Details |
| Mosunetuzumab | BTCT-4465A; RO-7030816; CD20-TBD; RG-7828 | Approved | Genentech Inc | Lunsumio | EU | Lymphoma, Follicular | Roche Registration Gmbh | 2022-06-03 | Lymphoma, B-Cell; Lymphoma, B-Cell, Marginal Zone; Lymphoma, Large B-Cell, Diffuse; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Follicular; Lupus Erythematosus, Systemic; Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Epcoritamab | GEN-3013; ABBV-GMAB-3013 | Approved | Genmab A/S, Abbvie Inc | Tepkinly, TEPKINLY, EPKINLY | United States | Lymphoma, Large B-Cell, Diffuse | Genmab Us Inc | 2023-05-19 | Lymphoma, B-Cell; Lymphoma, B-Cell, Marginal Zone; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Follicular; Lymphoma, Mantle-Cell; Primary mediastinal B cell lymphoma; Richter's Syndrome; Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Blinatumomab | BiTE-MT-103; bscCD19xCD3; AMG-103; MT-103; MEDI-538 | Approved | Micromet Inc | Blincyto, 倍利妥 | United States | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Amgen Inc | 2014-12-03 | Leukemia; Leukemia, Myelogenous, Chronic; Neoplasm, Residual; Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Multiple Myeloma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor T-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Philadelphia Chromosome; Lymphoma, Non-Hodgkin; Burkitt Lymphoma; Lymphoma; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| English Name | Research Code | Research Phase | Company | Indications | Clinical Trials |
|---|---|---|---|---|---|
| M-701 | M-701 | Phase 3 Clinical | Wuhan Yzy Biopharma Co Ltd | Ovarian Neoplasms; Solid tumours; Hydrothorax; Stomach Neoplasms; Neoplasms; Ascites; Colorectal Neoplasms; Pleural Effusion, Malignant; Carcinoma, Non-Small-Cell Lung | Details |
| ABBV-383 | TNB-383B; ABBV-383 | Phase 3 Clinical | Teneobio Inc, Abbvie Inc | Multiple Myeloma | Details |
| IMC-F106C | IMC-F106C; PRAME HLA-A02 | Phase 3 Clinical | Immunocore Ltd | Solid tumours; Melanoma | Details |
| Alnuctamab | CC-93269; EM-901; BMS-986349 | Phase 3 Clinical | Engmab Ag | Multiple Myeloma | Details |
| Catumaxomab | LP000 | Phase 3 Clinical | Trion Research, Neovii Biotech Gmbh | Ovarian Neoplasms; Stomach Neoplasms; Carcinoma; Neoplasms; Carcinoma, Ovarian Epithelial; Colonic Neoplasms; Urinary Bladder Neoplasms; Ascites; Breast Neoplasms | Details |
| Flotetuzumab | S-80880; MGD-006; RES-234 | Phase 2 Clinical | Macrogenics Inc | Leukemia, Myelogenous, Chronic; Hematologic Neoplasms; Leukemia; Leukemia, Hairy Cell; Mastocytosis, Systemic; Hodgkin Disease; Blastic Plasmacytoid Dendritic Cell Neoplasm; Precursor T-Cell Lymphoblastic Leukemia-Lymphoma; Neoplasms, Plasma Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Leukemia, Myeloid, Acute; Leukemia, Biphenotypic, Acute | Details |
| Anti-CD3/humanized 3F8 bispecific antibody-activated T lymphocytes (University of Virginia) | Phase 2 Clinical | University Of Virginia | Neuroblastoma | Details | |
| Imvotamab | IGM-2323 | Phase 2 Clinical | Igm Biosciences Inc | Lymphoma, B-Cell, Marginal Zone; Myositis; Lupus Erythematosus, Cutaneous; Arthritis, Rheumatoid; Lymphoma, Large B-Cell, Diffuse; Lupus Erythematosus, Systemic; Lymphoma, Mantle-Cell; Lymphoma, Follicular; Arthritis; Lymphoma, Non-Hodgkin | Details |
| Foralumab | TZLS-401; NI-0401/α-CD3; NI-0401 | Phase 2 Clinical | Novimmune Sa, Bristol-Myers Squibb Company | Respiratory Tract Infections; Diabetes Mellitus, Type 2; Metabolic Dysfunction-Associated Steatotic Liver Disease; Coronavirus Disease 2019 (COVID-19); Multiple Sclerosis, Chronic Progressive; Multiple Sclerosis; Alzheimer Disease; Crohn Disease | Details |
| GNR-084 | GNR-084 | Phase 2 Clinical | Generium Pharmaceuticals, Iontas | Precursor Cell Lymphoblastic Leukemia-Lymphoma; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details |
| Anti-CD3 and anti-HER2 BiTE-expressing T cell (MedImmune) | Phase 2 Clinical | University Of Michigan, Huazhong University Of Science And Technology, University Of Virginia Cancer Center, Medimmune | Breast Neoplasms | Details | |
| Vibecotamab | XmAb-14045 | Phase 2 Clinical | Xencor Inc | Leukemia, Myeloid; Myelodysplastic Syndromes; Blastic Plasmacytoid Dendritic Cell Neoplasm; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, Myeloid, Acute | Details |
| α/β CD3+/CD19+ cell depleted stem cell therapy (Mitchell Cairo) | Phase 2 Clinical | New York Medical College | Leukemia; Anemia; Thalassemia; Hodgkin Disease; Anemia, Aplastic; Thrombocytopenia; Lymphoma, Non-Hodgkin; Kostmann Syndrome; Anemia, Sickle Cell | Details | |
| MK-6070 | HPN-328; MK-6070 | Phase 2 Clinical | Harpoon Therapeutics | Small Cell Lung Carcinoma | Details |
| Cibisatamab | CEA-TCB; RG-7802; RO-6958688; CEA-CD3 TCB | Phase 2 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| CD3/CD19 neg allogeneic BMT (National Institute of Allergy and Infectious Diseases/University of Pittsburgh) | Phase 2 Clinical | National Institute Of Allergy And Infectious Diseases (Niaid), University Of Pittsburgh | Primary Immunodeficiency Diseases; Female Urogenital Diseases; Inflammation | Details | |
| HPN-536 | HPN-536 | Phase 2 Clinical | Harpoon Therapeutics | Neoplasms | Details |
| MBS-303 | MBS-303 | Phase 2 Clinical | Beijing Mabworks Biotech Co Ltd | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin | Details |
| HBM-7022 | HBM-7022; AZD-5863 | Phase 2 Clinical | Harbour Biomed | Esophageal Neoplasms; Stomach Neoplasms; Pancreatic Neoplasms; Neoplasms; Digestive System Neoplasms; Esophageal adenocarcinoma; Carcinoma, Pancreatic Ductal; Gastrointestinal Neoplasms | Details |
| TAK-280 | MVC-280; TAK-280 | Phase 2 Clinical | Maverick Therapeutics Inc | Solid tumours; Neoplasms; Neoplasm Metastasis | Details |
| GB-261 | GB-261 | Phase 2 Clinical | Genor Biopharma Co Ltd | Lymphoma, B-Cell; Leukemia, Lymphoid; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| REGN-5459 | REGN-5459 | Phase 2 Clinical | Multiple Myeloma; Renal Insufficiency, Chronic | Details | |
| IBI-3003 | IBI3003; IBI-3003 | Phase 2 Clinical | Innovent Biologics(Suzhou) Co Ltd | Multiple Myeloma | Details |
| MBS-314 | MBS-314 | Phase 2 Clinical | Beijing Mabworks Biotech Co Ltd | Multiple Myeloma | Details |
| RO7515629 | RO7515629; RO-7515629; RG-6353 | Phase 2 Clinical | F. Hoffmann-La Roche Ag | Ovarian Neoplasms; Carcinoma, Renal Cell; Pancreatic Neoplasms; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| BI-764532 | BI-764532; OBT620 | Phase 2 Clinical | C.H. Boehringer Sohn Ag & Co. Kg | Small Cell Lung Carcinoma; Neoplasms; Neuroendocrine Tumors; Carcinoma, Neuroendocrine; Glioma | Details |
| GEN3017 | GEN-3017 | Phase 2 Clinical | Genmab A/S | Hematologic Neoplasms; Hodgkin Disease; Lymphoma, Non-Hodgkin | Details |
| MP0533 | MP0533 | Phase 2 Clinical | Molecular Partners Ag | Leukemia, Myeloid, Acute | Details |
| CD30 biAb-AATC(The Medical College Of Wisconsin Nonprofit) | Phase 2 Clinical | The Medical College Of Wisconsin Nonprofit | Lymphoma, B-Cell; Leukemia; Hodgkin Disease; Lymphoma; Lymphoma, Large-Cell, Anaplastic; Lymphoma, T-Cell, Cutaneous | Details | |
| APVO-436 | APVO-436; APVO436 | Phase 2 Clinical | Aptevo | Myelodysplastic Syndromes; Leukemia, Myeloid, Acute | Details |
| Emfizatamab | GNC-038 | Phase 2 Clinical | SystImmune | Solid tumours; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Extranodal NK-T-Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin; Lymphoma, T-Cell; Central Nervous System Lymphoma | Details |
| GEN-1047 | GEN-1047 | Phase 2 Clinical | Genmab A/S | Carcinoma, Non-Small-Cell Lung | Details |
| REGN-4336 | REGN-4336 | Phase 2 Clinical | Prostatic Neoplasms, Castration-Resistant | Details | |
| CM-350 | CM-350 | Phase 2 Clinical | Keymed Biosciences Co Ltd | Solid tumours | Details |
| 1-A-46 | BR-110; 1A46; 1-A-46; CMG1A46; CMG1A-46; BR110 | Phase 2 Clinical | Chengdu Chimagen Biosciences Co Ltd, BioRay Pharmaceutical Co Ltd | Lymphoma, B-Cell; Neoplasms; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin | Details |
| LBL-034 | LBL-034 | Phase 2 Clinical | Nanjing Leads Biolabs Co Ltd | Neoplasms; Multiple Myeloma | Details |
| LBL-033 | LBL-033 | Phase 2 Clinical | Nanjing Leads Biolabs Co Ltd | Neoplasms | Details |
| CM-336(Connaught Biomedical Technology) | CM-336 | Phase 2 Clinical | Keymed Biosciences Co Ltd | Multiple Myeloma | Details |
| ICP-B02 | CM-355; ICP-B02 | Phase 2 Clinical | Beijing Tiannuo Jiancheng Pharmaceutical Technology Co Ltd, Keymed Biosciences Co Ltd | Hematologic Neoplasms; Hematoma; Lymphoma; Lymphoma, Non-Hodgkin | Details |
| SMET-12 | SMET-12 | Phase 2 Clinical | Zhejiang Shimai Pharmaceutical Co Ltd | Solid tumours; Carcinoma, Non-Small-Cell Lung | Details |
| QLS-31905 | QLS-31905 | Phase 2 Clinical | Qilu Pharmaceutical Co Ltd | Solid tumours; Neoplasms | Details |
| Nebratamig | GNC-035 | Phase 2 Clinical | Solid tumours; Hematologic Neoplasms; Breast Neoplasms; Metastatic breast cancer; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details | |
| TAK-186 | EGFR x CD3 COBRA; MVC-101; TAK-186 | Phase 2 Clinical | Takeda Pharmaceutical Co Ltd | Solid tumours; Squamous Cell Carcinoma of Head and Neck; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| EMB-06 | EMB-06; EMB06 | Phase 2 Clinical | Shanghai Epimab Biotherapeutics, Inc | Multiple Myeloma | Details |
| AZD-0486 | TNB-486; AZD-0486; AZD0486 | Phase 2 Clinical | Teneobio Inc | Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Follicular; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, Myeloid, Acute; Lymphoma, Non-Hodgkin | Details |
| BNT-142 | BNT-142 | Phase 2 Clinical | Biontech Se | Solid tumours | Details |
| CN-201 | CN-201 | Phase 2 Clinical | Curon Biopharmaceutical Ltd | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Lymphoma, Non-Hodgkin | Details |
| ZG006 | ZG006; ZG-006 | Phase 2 Clinical | Gensun Biopharma Inc, Suzhou Zelgen Biopharmaceuticals Co Ltd | Solid tumours; Neoplasms; Small Cell Lung Carcinoma; Carcinoma, Neuroendocrine | Details |
| EX-103 | EX-103; EX103 | Phase 2 Clinical | Guangzhou Excelmab Inc | Arthritis, Rheumatoid; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Anti-CD3 monoclonal antibody (Jinan Tiankang Biological Products) | Phase 2 Clinical | Jinan Tiankang Biological Products Co Ltd | Anemia, Aplastic | Details | |
| Cevostamab | RG-6160; BFCR-4350A; RO-7187797 | Phase 2 Clinical | Genentech Inc | Multiple Myeloma | Details |
| Anti-CD3/anti-EGFR -activated T cells (Barbara Ann Karmanos Cancer Institute) | Phase 2 Clinical | Barbara Ann Karmanos Cancer Institute, University Of Virginia | Glioblastoma; Pancreatic Neoplasms | Details | |
| Ubamatamab | REGN-4018 | Phase 2 Clinical | Ovarian Neoplasms; Peritoneal Neoplasms; Endometrial Neoplasms; Fallopian Tube Neoplasms | Details | |
| Anti-CD3-anti-HER2-activated T cells | Phase 2 Clinical | Transtarget | Prostatic Neoplasms, Castration-Resistant; Breast Neoplasms; Prostatic Neoplasms | Details | |
| JCAR-014 | JCAR-014; JCAR021 | Phase 2 Clinical | Fred Hutchinson Cancer Research Center, Memorial Sloan Kettering Cancer Center, Seattle Children'S Research Institute, Juno Therapeutics Inc | Lymphoma, B-Cell; Leukemia; Leukemia, Lymphoid; Hodgkin Disease; Lymphoma, Large B-Cell, Diffuse; Candidiasis; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Mantle-Cell; Primary mediastinal B cell lymphoma; Leukemia, B-Cell; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| A-319 | A-319 | Phase 1 Clinical | Evive Biotech Ltd | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell | Details |
| BCMA TriTAC | HPN-217 | Phase 1 Clinical | Harpoon Therapeutics, Abbvie Inc | Multiple Myeloma | Details |
| Membrane bound ligand T-SIGn virus | NG-348 | Phase 1 Clinical | Akamis Bio Ltd | Neoplasms | Details |
| NG-641 | EnAd-FAP-BiTE; EnAd-FAP-Tac; NG-641; NG-aFAP | Phase 1 Clinical | Akamis Bio Ltd, University Of Oxford | Squamous Cell Carcinoma of Head and Neck; Neoplasms, Glandular and Epithelial; Neoplasm Metastasis | Details |
| Vixtimotamab | T-564; AMV-564 | Phase 1 Clinical | Amphivena Therapeutics Inc | Solid tumours; Myelodysplastic Syndromes | Details |
| MGD-014 | MGD-014 | Phase 1 Clinical | Macrogenics Inc | HIV Infections | Details |
| JNJ-63898081 | JNJ-8081; JNJ-63898081 | Phase 1 Clinical | Johnson & Johnson Innovative Medicine | Neoplasms | Details |
| K-193 | K-193 | Phase 1 Clinical | Beijing Lvzhu Biological Technology Co Ltd | Lymphoma, B-Cell | Details |
| Anti-EGFR-bispecific antibody armed activated T-cell therapy (Memorial Sloan Kettering Cancer Center) | Phase 1 Clinical | Memorial Sloan Kettering Cancer Center | Pancreatic Neoplasms | Details | |
| Runimotamab | BTRC-4017A; RG-6194; RO-7227780 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours; Neoplasms | Details |
| Autologous T-cell therapy (anti-PSMA-CD3), Roger Williams Medical Center | Phase 1 Clinical | Roger Williams Medical Center | Prostatic Neoplasms | Details | |
| ONO-4685 | ONO-4685 | Phase 1 Clinical | Merus Nv | Psoriasis; Lymphoma, T-Cell; Plaque psoriasis | Details |
| WVT-078(Novartis Pharma) | WVT-078 | Phase 1 Clinical | Novartis Pharma Ag | Multiple Myeloma | Details |
| M-802(Wuhan Yzy Biopharma/CSPC Pharmaceutical) | M-802 | Phase 1 Clinical | Wuhan Yzy Biopharma Co Ltd, CSPC Pharmaceutical Group Ltd | Ovarian Neoplasms; Solid tumours; Stomach Neoplasms; Breast Neoplasms | Details |
| Emirodatamab | AMG-427 | Phase 1 Clinical | Amgen Inc | Leukemia, Myeloid, Acute | Details |
| JNJ-63709178 | JNJ-9178; CNTO-9958; JNJ-63709178 | Phase 1 Clinical | Johnson & Johnson Innovative Medicine, Genmab A/S | Leukemia, Myeloid, Acute | Details |
| SAR-442257 | SAR-442257 | Phase 1 Clinical | Sanofi | Neoplasms | Details |
| CC-1 | CC-1 | Phase 1 Clinical | University Of Tubingen, German Cancer Research Center (Dkfz) | Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms; Carcinoma, Squamous Cell | Details |
| Acapatamab | AMG-160 | Phase 1 Clinical | Amgen Inc | Prostatic Neoplasms, Castration-Resistant; Carcinoma, Non-Small-Cell Lung | Details |
| Pavurutamab | AMG-701 | Phase 1 Clinical | Amgen Inc | Multiple Myeloma | Details |
| Xaluritamig | AMG-509 | Phase 1 Clinical | Xencor Inc, Amgen Inc | Prostatic Neoplasms, Castration-Resistant | Details |
| CC-3 | CC-3 | Phase 1 Clinical | Eberhard Karls University Of Tubingen, Germany | Colorectal Neoplasms; Gastrointestinal Neoplasms | Details |
| JS-203 | JS-203 | Phase 1 Clinical | Shanghai Junshi Biological Engineering Co Ltd | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin | Details |
| BC-004 | BC-004 | Phase 1 Clinical | Shandong Buchang Pharmaceuticals Co Ltd | Solid tumours; Breast Neoplasms | Details |
| JY-016 | JY-016 | Phase 1 Clinical | Beijing Jingyitaixiang Technology Development Co Ltd, Beijing Eastern Biotech Co Ltd | Solid tumours; Colorectal Neoplasms; Lung Neoplasms | Details |
| EX-105 | EX-105; EX105 | Phase 1 Clinical | Guangzhou Excelmab Inc | Solid tumours | Details |
| EMB-07 | EMB-07 | Phase 1 Clinical | Shanghai Epimab Biotherapeutics, Inc | Solid tumours; Ovarian Neoplasms; Stomach Neoplasms; Neoplasms; Triple Negative Breast Neoplasms; Adenocarcinoma of Lung; Pancreatic Neoplasms; Urinary Bladder Neoplasms; Prostatic Neoplasms; Colorectal Neoplasms; Lymphoma; Uterine Neoplasms; Neoplasm Metastasis | Details |
| BA-3182 | BA-3182 | Phase 1 Clinical | Bioatla | Solid tumours; Adenocarcinoma | Details |
| BA-1202 | BA-1202 | Phase 1 Clinical | Solid tumours | Details | |
| CLN-978 | CLN-978 | Phase 1 Clinical | Adimab LLC | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin | Details |
| MGD-024 | MGD-024 | Phase 1 Clinical | Macrogenics Inc | Leukemia, Myelogenous, Chronic; Leukemia, Hairy Cell; Hodgkin Disease; Myelodysplastic Syndromes; Neoplasms; Blastic Plasmacytoid Dendritic Cell Neoplasm; Leukemia, B-Cell; Leukemia, Myeloid, Acute | Details |
| ARB-202 | ARB-202 | Phase 1 Clinical | Arbele Corp | Liver Neoplasms; Biliary Tract Neoplasms; Stomach Neoplasms; Pancreatic Neoplasms; Cholangiocarcinoma; Colorectal Neoplasms; Esophageal adenocarcinoma; Gastrointestinal Neoplasms | Details |
| CX-904 | CX-904 | Phase 1 Clinical | Amgen Inc, Cytomx Therapeutics Inc | Solid tumours; Neoplasms | Details |
| CLN-049 | CLN-049 | Phase 1 Clinical | University Of Tubingen, German Cancer Research Center (Dkfz) | Myelodysplastic Syndromes; Leukemia, Myeloid, Acute | Details |
| XmAb-819 | XmAb-819 | Phase 1 Clinical | Xencor Inc | Kidney Neoplasms; Carcinoma, Renal Cell | Details |
| JNJ-70218902 | JNJ-70218902 | Phase 1 Clinical | Johnson & Johnson Innovative Medicine | Neoplasms | Details |
| JNJ-67571244 | JNJ-67571244; JNJ-67371244; JNJ-1244 | Phase 1 Clinical | Johnson & Johnson | Myelodysplastic Syndromes; Leukemia, Myeloid, Acute | Details |
| SCTB-35 | SCTB35; SCTB-35 | Phase 1 Clinical | SinoCelltech Ltd | Lymphoma, B-Cell | Details |
| JNJ-87890387 | JNJ-87890387 | Phase 1 Clinical | Janssen Research & Development Llc | Solid tumours | Details |
| XmAb-541 | XmAb541; XmAb-541 | Phase 1 Clinical | Xencor Inc | Ovarian Neoplasms; Ovarian germ cell tumor; Germinoma; Endometrial Neoplasms; Neoplasms, Germ Cell and Embryonal | Details |
| [89Zr]Zr-BI-764532 | Phase 1 Clinical | C.H. Boehringer Sohn Ag & Co. Kg, Boehringer Ingelheim Gmbh | Small Cell Lung Carcinoma; Carcinoma, Neuroendocrine | Details | |
| TGI-6 | TGI-6 | Phase 1 Clinical | Hefei TG ImmunoPharma Co Ltd | Solid tumours; Colorectal Neoplasms | Details |
| SIM-0500 | SIM-0500; SIM0500 | Phase 1 Clinical | Hainan Xiansheng Re Ming Pharmaceutical Co Ltd | Bone Marrow Neoplasms; Multiple Myeloma | Details |
| Recombinant anti BCMA/CD3 bispecific antibody(Hualan Genetic Engineering) | Phase 1 Clinical | Hualan Genetic Engineering (Henan) Co Ltd | Multiple Myeloma | Details | |
| Oncolytic Virus R130(Yunying Medical) | R-130-OV; R130; R-130 | Phase 1 Clinical | Shanghai Yunying Medical Technology Co Ltd | Breast Neoplasms; Carcinoma, Non-Small-Cell Lung; Gastrointestinal Neoplasms; Melanoma; Laryngeal Neoplasms; Pharyngeal Neoplasms; Lung Neoplasms; Fallopian Tube Neoplasms; Colorectal Neoplasms; Genital Neoplasms, Female; Bone Neoplasms; Peritoneal Neoplasms; Sarcoma; Osteosarcoma; Head and Neck Neoplasms; Brain Neoplasms; Otorhinolaryngologic Neoplasms; Small Cell Lung Carcinoma; Pancreatic Neoplasms; Nose Neoplasms; Stomach Neoplasms; Esophageal Neoplasms; Carcinoma; Carcinoma, Bronchogenic; Liver Neoplasms; Ovarian Neoplasms; Kidney Neoplasms | Details |
| ASP2074 | ASP-2074; ASP2074 | Phase 1 Clinical | Astellas Pharma Global Development Inc | Solid tumours | Details |
| OBT620 | OBT620 | Phase 1 Clinical | Boehringer Ingelheim Gmbh, Oxford Biotherapeutics Ltd | Small Cell Lung Carcinoma | Details |
| CBA-1535 | CBA-1535 | Phase 1 Clinical | Chiome Bioscience Inc | Small Cell Lung Carcinoma; Triple Negative Breast Neoplasms; Mesothelioma; Carcinoma, Non-Small-Cell Lung | Details |
| JANX-008 | EGFR-TRACTr; JANX008; JANX-008 | Phase 1 Clinical | Janux Therapeutics Inc | Solid tumours; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| JANX-007 | JANX-007; PSMA-TRACTr | Phase 1 Clinical | Janux Therapeutics Inc | Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms | Details |
| TQB-2934 | TQB2934; TQB-2934 | Phase 1 Clinical | Nanjing Shunxin Pharmaceuticals Co Ltd Of Chiatai Tianqing Pharmaceutical Group | Multiple Myeloma | Details |
| JNJ-80948543 | JNJ-80948543 | Phase 1 Clinical | Janssen Research & Development Llc | Lymphoma, B-Cell; Neoplasms; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| PIT-565 | PIT-565 | Phase 1 Clinical | Novartis Pharma Ag | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lupus Erythematosus, Systemic; Leukemia, Myeloid, Acute | Details |
| CC-312 | CC-312 | Phase 1 Clinical | CytoCares (Shanghai) Inc | Hematologic Neoplasms; Lymphoma, B-Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| ASP-2138 | ASP-2138 | Phase 1 Clinical | Astellas Pharma Global Development Inc, Xencor Inc | Stomach Neoplasms; Esophageal Neoplasms; Pancreatic Neoplasms | Details |
| ISB-2001 | ISB-2001 | Phase 1 Clinical | Ichnos Sciences Sa | Multiple Myeloma | Details |
| GR-1901 | GR1901; GR-1901 | Phase 1 Clinical | Chongqing Zhixiang Jintai Biopharmaceutical Co Ltd, Genrix (Shanghai) Biopharmaceutical Co Ltd | Leukemia, Myeloid, Acute | Details |
| AMG-794 | AMG-794 | Phase 1 Clinical | Amgen Inc | Carcinoma, Ovarian Epithelial; Carcinoma, Non-Small-Cell Lung | Details |
| QLS-31904 | QLS31904; QLS-31904 | Phase 1 Clinical | Qilu Pharmaceutical Co Ltd | Solid tumours | Details |
| YK-012 | YK012 | Phase 1 Clinical | Lymphoma, B-Cell | Details | |
| RO-7428731 | RO-7428731; RG-6156 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Glioblastoma | Details |
| GBR-1342 | GBR-1342; ISB-1342 | Phase 1 Clinical | Glenmark Pharmaceuticals Ltd | Multiple Myeloma | Details |
| Recombinant humanized anti-CD19/CD3 bispecific antibody(New Time Pharmaceutical) | LNF-1904 | Phase 1 Clinical | Shandong New Time Pharmaceutical Co Ltd | Lymphoma, B-Cell; Leukemia, B-Cell | Details |
| XmAb-968 | XmAb968 | Phase 1 Clinical | Xencor Inc | Hematologic Neoplasms; Leukemia, Promyelocytic, Acute | Details |
| GR-1803 | GR-1803 | Phase 1 Clinical | Chongqing Zhixiang Jintai Biopharmaceutical Co Ltd, Genrix (Shanghai) Biopharmaceutical Co Ltd | Multiple Myeloma | Details |
| XmAb-18968 | XmAb-18968 | Phase 1 Clinical | The Medical College Of Wisconsin Nonprofit | Leukemia, Myeloid, Acute | Details |
| SQZ-622(Novartis Pharma) | SQZ-622 | Phase 1 Clinical | Novartis Pharma Ag | Leukemia, Myeloid, Acute | Details |
| Forimtamig | RG-6234 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Multiple Myeloma | Details |
| RG-6232 | RG-6232 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Melanoma | Details |
| RG-6007 | RO-7283420; RG-6007 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Leukemia, Myeloid, Acute | Details |
| SAR-443216 | SAR-443216 | Phase 1 Clinical | Sanofi | Solid tumours; Stomach Neoplasms; Neoplasms; Breast Neoplasms; Lung Neoplasms | Details |
| Recombinant oncolytic type II herpes simplex virus (Binhui Biopharmaceutical) | BS-006; oHSV2-BiTEs | Phase 1 Clinical | Wuhan Binhui Biotechnology Co Ltd | Solid tumours; Neoplasms; Melanoma; Uterine Cervical Neoplasms | Details |
| JNJ-78306358 | JNJ-78306358; JNJ-6358 | Phase 1 Clinical | Janssen Research & Development Llc | Solid tumours | Details |
| F-182112 | F-182112; F182112 | Phase 1 Clinical | Shandong New Time Pharmaceutical Co Ltd | Multiple Myeloma | Details |
| TNB-585 | TNB-585; AMG-340 | Phase 1 Clinical | Teneobio Inc | Prostatic Neoplasms, Castration-Resistant | Details |
| BC-3448 | BC3448; BC-3448 | Phase 1 Clinical | Solid tumours | Details | |
| IBI-389 | IBI-389 | Phase 1 Clinical | Innovent Biologics(Suzhou) Co Ltd | Solid tumours; Neoplasms | Details |
| hEGFRvIII-CD3 Bi-scFv | Phase 1 Clinical | Duke University | Glioblastoma; Glioma | Details | |
| JNJ-78278343 | Phase 1 Clinical | Janssen Research & Development Llc | Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms | Details | |
| TQB-2825 | TQB-2825 | Phase 1 Clinical | Wuxi Biologics Co Ltd | Hematologic Neoplasms | Details |
| RGV-004 | RGV-004 | Phase 1 Clinical | Hangzhou Rongu Biotechnology Co Ltd | Lymphoma, B-Cell | Details |
| Recombinant humanized anti-BCMA/CD3 bispecific antibody(New Time Pharmaceutical) | Phase 1 Clinical | Shandong New Time Pharmaceutical Co Ltd | Multiple Myeloma | Details | |
| NVG-111 | NVG-111 | Phase 1 Clinical | NovalGen Ltd | Lymphoma, Large B-Cell, Diffuse; Lymphoma, Mantle-Cell; Lymphoma, Follicular; Melanoma; Carcinoma, Non-Small-Cell Lung; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| BI-765049 | BI-765049; OBT-624 | Phase 1 Clinical | Oxford Biotherapeutics Ltd | Solid tumours | Details |
| GNC-039 | GNC-039 | Phase 1 Clinical | Solid tumours; Hematologic Neoplasms; Glioma; Neoplasm Metastasis | Details | |
| RO-7425781 | RO-7425781 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Multiple Myeloma | Details |
| RO-7293583 | RO-7293583 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Skin Melanoma; Uveal melanoma; Melanoma | Details |
| Rezetamig | JNJ-8780; JNJ75348780; JNJ-75348780 | Phase 1 Clinical | Teneobio Inc | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Y-150(Wuhan Yzy Biopharma/CSPC Pharmaceutical) | Y-150; Y150 | Phase 1 Clinical | Wuhan Yzy Biopharma Co Ltd, CSPC Pharmaceutical Group Ltd | Multiple Myeloma | Details |
| EX-101 | EX-101; EX101 | Phase 1 Clinical | Guangzhou Excelmab Inc | Solid tumours | Details |
| Plamotamab | XmAb-13676 | Phase 1 Clinical | Xencor Inc | Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| A-337 | A-337 | Phase 1 Clinical | Evive Biotech Ltd | Solid tumours; Neoplasms | Details |
| CCW-702 | CCW-702 | Phase 1 Clinical | The Scripps Research Institute Inc, Abbvie Inc | Prostatic Neoplasms | Details |
| Tepoditamab | MCLA-117 | Phase 1 Clinical | Pharmaceutical Research Associates, Institute Gustave-Roussy, Merus Nv, Vu University Medical Center, Lgc | Leukemia, Myeloid, Acute | Details |
| ERY-974 | ERY-974 | Phase 1 Clinical | Chugai Pharmaceutical Co Ltd | Solid tumours; Carcinoma, Hepatocellular | Details |
| InHeAb-01 | InHeAb-01; bsAB | Clinical | University Hospital Tuebingen | Neoplasms | Details |
| M-701 | M-701 | Phase 3 Clinical | Wuhan Yzy Biopharma Co Ltd | Ovarian Neoplasms; Solid tumours; Hydrothorax; Stomach Neoplasms; Neoplasms; Ascites; Colorectal Neoplasms; Pleural Effusion, Malignant; Carcinoma, Non-Small-Cell Lung | Details |
| ABBV-383 | TNB-383B; ABBV-383 | Phase 3 Clinical | Teneobio Inc, Abbvie Inc | Multiple Myeloma | Details |
| IMC-F106C | IMC-F106C; PRAME HLA-A02 | Phase 3 Clinical | Immunocore Ltd | Solid tumours; Melanoma | Details |
| Alnuctamab | CC-93269; EM-901; BMS-986349 | Phase 3 Clinical | Engmab Ag | Multiple Myeloma | Details |
| Catumaxomab | LP000 | Phase 3 Clinical | Trion Research, Neovii Biotech Gmbh | Ovarian Neoplasms; Stomach Neoplasms; Carcinoma; Neoplasms; Carcinoma, Ovarian Epithelial; Colonic Neoplasms; Urinary Bladder Neoplasms; Ascites; Breast Neoplasms | Details |
| Flotetuzumab | S-80880; MGD-006; RES-234 | Phase 2 Clinical | Macrogenics Inc | Leukemia, Myelogenous, Chronic; Hematologic Neoplasms; Leukemia; Leukemia, Hairy Cell; Mastocytosis, Systemic; Hodgkin Disease; Blastic Plasmacytoid Dendritic Cell Neoplasm; Precursor T-Cell Lymphoblastic Leukemia-Lymphoma; Neoplasms, Plasma Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Leukemia, Myeloid, Acute; Leukemia, Biphenotypic, Acute | Details |
| Anti-CD3/humanized 3F8 bispecific antibody-activated T lymphocytes (University of Virginia) | Phase 2 Clinical | University Of Virginia | Neuroblastoma | Details | |
| Imvotamab | IGM-2323 | Phase 2 Clinical | Igm Biosciences Inc | Lymphoma, B-Cell, Marginal Zone; Myositis; Lupus Erythematosus, Cutaneous; Arthritis, Rheumatoid; Lymphoma, Large B-Cell, Diffuse; Lupus Erythematosus, Systemic; Lymphoma, Mantle-Cell; Lymphoma, Follicular; Arthritis; Lymphoma, Non-Hodgkin | Details |
| Foralumab | TZLS-401; NI-0401/α-CD3; NI-0401 | Phase 2 Clinical | Novimmune Sa, Bristol-Myers Squibb Company | Respiratory Tract Infections; Diabetes Mellitus, Type 2; Metabolic Dysfunction-Associated Steatotic Liver Disease; Coronavirus Disease 2019 (COVID-19); Multiple Sclerosis, Chronic Progressive; Multiple Sclerosis; Alzheimer Disease; Crohn Disease | Details |
| GNR-084 | GNR-084 | Phase 2 Clinical | Generium Pharmaceuticals, Iontas | Precursor Cell Lymphoblastic Leukemia-Lymphoma; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma | Details |
| Anti-CD3 and anti-HER2 BiTE-expressing T cell (MedImmune) | Phase 2 Clinical | University Of Michigan, Huazhong University Of Science And Technology, University Of Virginia Cancer Center, Medimmune | Breast Neoplasms | Details | |
| Vibecotamab | XmAb-14045 | Phase 2 Clinical | Xencor Inc | Leukemia, Myeloid; Myelodysplastic Syndromes; Blastic Plasmacytoid Dendritic Cell Neoplasm; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, Myeloid, Acute | Details |
| α/β CD3+/CD19+ cell depleted stem cell therapy (Mitchell Cairo) | Phase 2 Clinical | New York Medical College | Leukemia; Anemia; Thalassemia; Hodgkin Disease; Anemia, Aplastic; Thrombocytopenia; Lymphoma, Non-Hodgkin; Kostmann Syndrome; Anemia, Sickle Cell | Details | |
| MK-6070 | HPN-328; MK-6070 | Phase 2 Clinical | Harpoon Therapeutics | Small Cell Lung Carcinoma | Details |
| Cibisatamab | CEA-TCB; RG-7802; RO-6958688; CEA-CD3 TCB | Phase 2 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| CD3/CD19 neg allogeneic BMT (National Institute of Allergy and Infectious Diseases/University of Pittsburgh) | Phase 2 Clinical | National Institute Of Allergy And Infectious Diseases (Niaid), University Of Pittsburgh | Primary Immunodeficiency Diseases; Female Urogenital Diseases; Inflammation | Details | |
| HPN-536 | HPN-536 | Phase 2 Clinical | Harpoon Therapeutics | Neoplasms | Details |
| MBS-303 | MBS-303 | Phase 2 Clinical | Beijing Mabworks Biotech Co Ltd | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin | Details |
| HBM-7022 | HBM-7022; AZD-5863 | Phase 2 Clinical | Harbour Biomed | Esophageal Neoplasms; Stomach Neoplasms; Pancreatic Neoplasms; Neoplasms; Digestive System Neoplasms; Esophageal adenocarcinoma; Carcinoma, Pancreatic Ductal; Gastrointestinal Neoplasms | Details |
| TAK-280 | MVC-280; TAK-280 | Phase 2 Clinical | Maverick Therapeutics Inc | Solid tumours; Neoplasms; Neoplasm Metastasis | Details |
| GB-261 | GB-261 | Phase 2 Clinical | Genor Biopharma Co Ltd | Lymphoma, B-Cell; Leukemia, Lymphoid; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| REGN-5459 | REGN-5459 | Phase 2 Clinical | Multiple Myeloma; Renal Insufficiency, Chronic | Details | |
| IBI-3003 | IBI3003; IBI-3003 | Phase 2 Clinical | Innovent Biologics(Suzhou) Co Ltd | Multiple Myeloma | Details |
| MBS-314 | MBS-314 | Phase 2 Clinical | Beijing Mabworks Biotech Co Ltd | Multiple Myeloma | Details |
| RO7515629 | RO7515629; RO-7515629; RG-6353 | Phase 2 Clinical | F. Hoffmann-La Roche Ag | Ovarian Neoplasms; Carcinoma, Renal Cell; Pancreatic Neoplasms; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| BI-764532 | BI-764532; OBT620 | Phase 2 Clinical | C.H. Boehringer Sohn Ag & Co. Kg | Small Cell Lung Carcinoma; Neoplasms; Neuroendocrine Tumors; Carcinoma, Neuroendocrine; Glioma | Details |
| GEN3017 | GEN-3017 | Phase 2 Clinical | Genmab A/S | Hematologic Neoplasms; Hodgkin Disease; Lymphoma, Non-Hodgkin | Details |
| MP0533 | MP0533 | Phase 2 Clinical | Molecular Partners Ag | Leukemia, Myeloid, Acute | Details |
| CD30 biAb-AATC(The Medical College Of Wisconsin Nonprofit) | Phase 2 Clinical | The Medical College Of Wisconsin Nonprofit | Lymphoma, B-Cell; Leukemia; Hodgkin Disease; Lymphoma; Lymphoma, Large-Cell, Anaplastic; Lymphoma, T-Cell, Cutaneous | Details | |
| APVO-436 | APVO-436; APVO436 | Phase 2 Clinical | Aptevo | Myelodysplastic Syndromes; Leukemia, Myeloid, Acute | Details |
| Emfizatamab | GNC-038 | Phase 2 Clinical | SystImmune | Solid tumours; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Extranodal NK-T-Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin; Lymphoma, T-Cell; Central Nervous System Lymphoma | Details |
| GEN-1047 | GEN-1047 | Phase 2 Clinical | Genmab A/S | Carcinoma, Non-Small-Cell Lung | Details |
| REGN-4336 | REGN-4336 | Phase 2 Clinical | Prostatic Neoplasms, Castration-Resistant | Details | |
| CM-350 | CM-350 | Phase 2 Clinical | Keymed Biosciences Co Ltd | Solid tumours | Details |
| 1-A-46 | BR-110; 1A46; 1-A-46; CMG1A46; CMG1A-46; BR110 | Phase 2 Clinical | Chengdu Chimagen Biosciences Co Ltd, BioRay Pharmaceutical Co Ltd | Lymphoma, B-Cell; Neoplasms; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin | Details |
| LBL-034 | LBL-034 | Phase 2 Clinical | Nanjing Leads Biolabs Co Ltd | Neoplasms; Multiple Myeloma | Details |
| LBL-033 | LBL-033 | Phase 2 Clinical | Nanjing Leads Biolabs Co Ltd | Neoplasms | Details |
| CM-336(Connaught Biomedical Technology) | CM-336 | Phase 2 Clinical | Keymed Biosciences Co Ltd | Multiple Myeloma | Details |
| ICP-B02 | CM-355; ICP-B02 | Phase 2 Clinical | Beijing Tiannuo Jiancheng Pharmaceutical Technology Co Ltd, Keymed Biosciences Co Ltd | Hematologic Neoplasms; Hematoma; Lymphoma; Lymphoma, Non-Hodgkin | Details |
| SMET-12 | SMET-12 | Phase 2 Clinical | Zhejiang Shimai Pharmaceutical Co Ltd | Solid tumours; Carcinoma, Non-Small-Cell Lung | Details |
| QLS-31905 | QLS-31905 | Phase 2 Clinical | Qilu Pharmaceutical Co Ltd | Solid tumours; Neoplasms | Details |
| Nebratamig | GNC-035 | Phase 2 Clinical | Solid tumours; Hematologic Neoplasms; Breast Neoplasms; Metastatic breast cancer; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details | |
| TAK-186 | EGFR x CD3 COBRA; MVC-101; TAK-186 | Phase 2 Clinical | Takeda Pharmaceutical Co Ltd | Solid tumours; Squamous Cell Carcinoma of Head and Neck; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| EMB-06 | EMB-06; EMB06 | Phase 2 Clinical | Shanghai Epimab Biotherapeutics, Inc | Multiple Myeloma | Details |
| AZD-0486 | TNB-486; AZD-0486; AZD0486 | Phase 2 Clinical | Teneobio Inc | Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Follicular; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, Myeloid, Acute; Lymphoma, Non-Hodgkin | Details |
| BNT-142 | BNT-142 | Phase 2 Clinical | Biontech Se | Solid tumours | Details |
| CN-201 | CN-201 | Phase 2 Clinical | Curon Biopharmaceutical Ltd | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell; Lymphoma, Non-Hodgkin | Details |
| ZG006 | ZG006; ZG-006 | Phase 2 Clinical | Gensun Biopharma Inc, Suzhou Zelgen Biopharmaceuticals Co Ltd | Solid tumours; Neoplasms; Small Cell Lung Carcinoma; Carcinoma, Neuroendocrine | Details |
| EX-103 | EX-103; EX103 | Phase 2 Clinical | Guangzhou Excelmab Inc | Arthritis, Rheumatoid; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Anti-CD3 monoclonal antibody (Jinan Tiankang Biological Products) | Phase 2 Clinical | Jinan Tiankang Biological Products Co Ltd | Anemia, Aplastic | Details | |
| Cevostamab | RG-6160; BFCR-4350A; RO-7187797 | Phase 2 Clinical | Genentech Inc | Multiple Myeloma | Details |
| Anti-CD3/anti-EGFR -activated T cells (Barbara Ann Karmanos Cancer Institute) | Phase 2 Clinical | Barbara Ann Karmanos Cancer Institute, University Of Virginia | Glioblastoma; Pancreatic Neoplasms | Details | |
| Ubamatamab | REGN-4018 | Phase 2 Clinical | Ovarian Neoplasms; Peritoneal Neoplasms; Endometrial Neoplasms; Fallopian Tube Neoplasms | Details | |
| Anti-CD3-anti-HER2-activated T cells | Phase 2 Clinical | Transtarget | Prostatic Neoplasms, Castration-Resistant; Breast Neoplasms; Prostatic Neoplasms | Details | |
| JCAR-014 | JCAR-014; JCAR021 | Phase 2 Clinical | Fred Hutchinson Cancer Research Center, Memorial Sloan Kettering Cancer Center, Seattle Children'S Research Institute, Juno Therapeutics Inc | Lymphoma, B-Cell; Leukemia; Leukemia, Lymphoid; Hodgkin Disease; Lymphoma, Large B-Cell, Diffuse; Candidiasis; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Mantle-Cell; Primary mediastinal B cell lymphoma; Leukemia, B-Cell; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| A-319 | A-319 | Phase 1 Clinical | Evive Biotech Ltd | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Leukemia, B-Cell | Details |
| BCMA TriTAC | HPN-217 | Phase 1 Clinical | Harpoon Therapeutics, Abbvie Inc | Multiple Myeloma | Details |
| Membrane bound ligand T-SIGn virus | NG-348 | Phase 1 Clinical | Akamis Bio Ltd | Neoplasms | Details |
| NG-641 | EnAd-FAP-BiTE; EnAd-FAP-Tac; NG-641; NG-aFAP | Phase 1 Clinical | Akamis Bio Ltd, University Of Oxford | Squamous Cell Carcinoma of Head and Neck; Neoplasms, Glandular and Epithelial; Neoplasm Metastasis | Details |
| Vixtimotamab | T-564; AMV-564 | Phase 1 Clinical | Amphivena Therapeutics Inc | Solid tumours; Myelodysplastic Syndromes | Details |
| MGD-014 | MGD-014 | Phase 1 Clinical | Macrogenics Inc | HIV Infections | Details |
| JNJ-63898081 | JNJ-8081; JNJ-63898081 | Phase 1 Clinical | Johnson & Johnson Innovative Medicine | Neoplasms | Details |
| K-193 | K-193 | Phase 1 Clinical | Beijing Lvzhu Biological Technology Co Ltd | Lymphoma, B-Cell | Details |
| Anti-EGFR-bispecific antibody armed activated T-cell therapy (Memorial Sloan Kettering Cancer Center) | Phase 1 Clinical | Memorial Sloan Kettering Cancer Center | Pancreatic Neoplasms | Details | |
| Runimotamab | BTRC-4017A; RG-6194; RO-7227780 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Solid tumours; Neoplasms | Details |
| Autologous T-cell therapy (anti-PSMA-CD3), Roger Williams Medical Center | Phase 1 Clinical | Roger Williams Medical Center | Prostatic Neoplasms | Details | |
| ONO-4685 | ONO-4685 | Phase 1 Clinical | Merus Nv | Psoriasis; Lymphoma, T-Cell; Plaque psoriasis | Details |
| WVT-078(Novartis Pharma) | WVT-078 | Phase 1 Clinical | Novartis Pharma Ag | Multiple Myeloma | Details |
| M-802(Wuhan Yzy Biopharma/CSPC Pharmaceutical) | M-802 | Phase 1 Clinical | Wuhan Yzy Biopharma Co Ltd, CSPC Pharmaceutical Group Ltd | Ovarian Neoplasms; Solid tumours; Stomach Neoplasms; Breast Neoplasms | Details |
| Emirodatamab | AMG-427 | Phase 1 Clinical | Amgen Inc | Leukemia, Myeloid, Acute | Details |
| JNJ-63709178 | JNJ-9178; CNTO-9958; JNJ-63709178 | Phase 1 Clinical | Johnson & Johnson Innovative Medicine, Genmab A/S | Leukemia, Myeloid, Acute | Details |
| SAR-442257 | SAR-442257 | Phase 1 Clinical | Sanofi | Neoplasms | Details |
| CC-1 | CC-1 | Phase 1 Clinical | University Of Tubingen, German Cancer Research Center (Dkfz) | Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms; Carcinoma, Squamous Cell | Details |
| Acapatamab | AMG-160 | Phase 1 Clinical | Amgen Inc | Prostatic Neoplasms, Castration-Resistant; Carcinoma, Non-Small-Cell Lung | Details |
| Pavurutamab | AMG-701 | Phase 1 Clinical | Amgen Inc | Multiple Myeloma | Details |
| Xaluritamig | AMG-509 | Phase 1 Clinical | Xencor Inc, Amgen Inc | Prostatic Neoplasms, Castration-Resistant | Details |
| CC-3 | CC-3 | Phase 1 Clinical | Eberhard Karls University Of Tubingen, Germany | Colorectal Neoplasms; Gastrointestinal Neoplasms | Details |
| JS-203 | JS-203 | Phase 1 Clinical | Shanghai Junshi Biological Engineering Co Ltd | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin | Details |
| BC-004 | BC-004 | Phase 1 Clinical | Shandong Buchang Pharmaceuticals Co Ltd | Solid tumours; Breast Neoplasms | Details |
| JY-016 | JY-016 | Phase 1 Clinical | Beijing Jingyitaixiang Technology Development Co Ltd, Beijing Eastern Biotech Co Ltd | Solid tumours; Colorectal Neoplasms; Lung Neoplasms | Details |
| EX-105 | EX-105; EX105 | Phase 1 Clinical | Guangzhou Excelmab Inc | Solid tumours | Details |
| EMB-07 | EMB-07 | Phase 1 Clinical | Shanghai Epimab Biotherapeutics, Inc | Solid tumours; Ovarian Neoplasms; Stomach Neoplasms; Neoplasms; Triple Negative Breast Neoplasms; Adenocarcinoma of Lung; Pancreatic Neoplasms; Urinary Bladder Neoplasms; Prostatic Neoplasms; Colorectal Neoplasms; Lymphoma; Uterine Neoplasms; Neoplasm Metastasis | Details |
| BA-3182 | BA-3182 | Phase 1 Clinical | Bioatla | Solid tumours; Adenocarcinoma | Details |
| BA-1202 | BA-1202 | Phase 1 Clinical | Solid tumours | Details | |
| CLN-978 | CLN-978 | Phase 1 Clinical | Adimab LLC | Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin | Details |
| MGD-024 | MGD-024 | Phase 1 Clinical | Macrogenics Inc | Leukemia, Myelogenous, Chronic; Leukemia, Hairy Cell; Hodgkin Disease; Myelodysplastic Syndromes; Neoplasms; Blastic Plasmacytoid Dendritic Cell Neoplasm; Leukemia, B-Cell; Leukemia, Myeloid, Acute | Details |
| ARB-202 | ARB-202 | Phase 1 Clinical | Arbele Corp | Liver Neoplasms; Biliary Tract Neoplasms; Stomach Neoplasms; Pancreatic Neoplasms; Cholangiocarcinoma; Colorectal Neoplasms; Esophageal adenocarcinoma; Gastrointestinal Neoplasms | Details |
| CX-904 | CX-904 | Phase 1 Clinical | Amgen Inc, Cytomx Therapeutics Inc | Solid tumours; Neoplasms | Details |
| CLN-049 | CLN-049 | Phase 1 Clinical | University Of Tubingen, German Cancer Research Center (Dkfz) | Myelodysplastic Syndromes; Leukemia, Myeloid, Acute | Details |
| XmAb-819 | XmAb-819 | Phase 1 Clinical | Xencor Inc | Kidney Neoplasms; Carcinoma, Renal Cell | Details |
| JNJ-70218902 | JNJ-70218902 | Phase 1 Clinical | Johnson & Johnson Innovative Medicine | Neoplasms | Details |
| JNJ-67571244 | JNJ-67571244; JNJ-67371244; JNJ-1244 | Phase 1 Clinical | Johnson & Johnson | Myelodysplastic Syndromes; Leukemia, Myeloid, Acute | Details |
| SCTB-35 | SCTB35; SCTB-35 | Phase 1 Clinical | SinoCelltech Ltd | Lymphoma, B-Cell | Details |
| JNJ-87890387 | JNJ-87890387 | Phase 1 Clinical | Janssen Research & Development Llc | Solid tumours | Details |
| XmAb-541 | XmAb541; XmAb-541 | Phase 1 Clinical | Xencor Inc | Ovarian Neoplasms; Ovarian germ cell tumor; Germinoma; Endometrial Neoplasms; Neoplasms, Germ Cell and Embryonal | Details |
| [89Zr]Zr-BI-764532 | Phase 1 Clinical | C.H. Boehringer Sohn Ag & Co. Kg, Boehringer Ingelheim Gmbh | Small Cell Lung Carcinoma; Carcinoma, Neuroendocrine | Details | |
| TGI-6 | TGI-6 | Phase 1 Clinical | Hefei TG ImmunoPharma Co Ltd | Solid tumours; Colorectal Neoplasms | Details |
| SIM-0500 | SIM-0500; SIM0500 | Phase 1 Clinical | Hainan Xiansheng Re Ming Pharmaceutical Co Ltd | Bone Marrow Neoplasms; Multiple Myeloma | Details |
| Recombinant anti BCMA/CD3 bispecific antibody(Hualan Genetic Engineering) | Phase 1 Clinical | Hualan Genetic Engineering (Henan) Co Ltd | Multiple Myeloma | Details | |
| Oncolytic Virus R130(Yunying Medical) | R-130-OV; R130; R-130 | Phase 1 Clinical | Shanghai Yunying Medical Technology Co Ltd | Breast Neoplasms; Carcinoma, Non-Small-Cell Lung; Gastrointestinal Neoplasms; Melanoma; Laryngeal Neoplasms; Pharyngeal Neoplasms; Lung Neoplasms; Fallopian Tube Neoplasms; Colorectal Neoplasms; Genital Neoplasms, Female; Bone Neoplasms; Peritoneal Neoplasms; Sarcoma; Osteosarcoma; Head and Neck Neoplasms; Brain Neoplasms; Otorhinolaryngologic Neoplasms; Small Cell Lung Carcinoma; Pancreatic Neoplasms; Nose Neoplasms; Stomach Neoplasms; Esophageal Neoplasms; Carcinoma; Carcinoma, Bronchogenic; Liver Neoplasms; Ovarian Neoplasms; Kidney Neoplasms | Details |
| ASP2074 | ASP-2074; ASP2074 | Phase 1 Clinical | Astellas Pharma Global Development Inc | Solid tumours | Details |
| OBT620 | OBT620 | Phase 1 Clinical | Boehringer Ingelheim Gmbh, Oxford Biotherapeutics Ltd | Small Cell Lung Carcinoma | Details |
| CBA-1535 | CBA-1535 | Phase 1 Clinical | Chiome Bioscience Inc | Small Cell Lung Carcinoma; Triple Negative Breast Neoplasms; Mesothelioma; Carcinoma, Non-Small-Cell Lung | Details |
| JANX-008 | EGFR-TRACTr; JANX008; JANX-008 | Phase 1 Clinical | Janux Therapeutics Inc | Solid tumours; Squamous Cell Carcinoma of Head and Neck; Carcinoma, Renal Cell; Colorectal Neoplasms; Carcinoma, Non-Small-Cell Lung | Details |
| JANX-007 | JANX-007; PSMA-TRACTr | Phase 1 Clinical | Janux Therapeutics Inc | Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms | Details |
| TQB-2934 | TQB2934; TQB-2934 | Phase 1 Clinical | Nanjing Shunxin Pharmaceuticals Co Ltd Of Chiatai Tianqing Pharmaceutical Group | Multiple Myeloma | Details |
| JNJ-80948543 | JNJ-80948543 | Phase 1 Clinical | Janssen Research & Development Llc | Lymphoma, B-Cell; Neoplasms; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| PIT-565 | PIT-565 | Phase 1 Clinical | Novartis Pharma Ag | Lymphoma, B-Cell; Precursor B-Cell Lymphoblastic Leukemia-Lymphoma; Lupus Erythematosus, Systemic; Leukemia, Myeloid, Acute | Details |
| CC-312 | CC-312 | Phase 1 Clinical | CytoCares (Shanghai) Inc | Hematologic Neoplasms; Lymphoma, B-Cell; Precursor Cell Lymphoblastic Leukemia-Lymphoma; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| ASP-2138 | ASP-2138 | Phase 1 Clinical | Astellas Pharma Global Development Inc, Xencor Inc | Stomach Neoplasms; Esophageal Neoplasms; Pancreatic Neoplasms | Details |
| ISB-2001 | ISB-2001 | Phase 1 Clinical | Ichnos Sciences Sa | Multiple Myeloma | Details |
| GR-1901 | GR1901; GR-1901 | Phase 1 Clinical | Chongqing Zhixiang Jintai Biopharmaceutical Co Ltd, Genrix (Shanghai) Biopharmaceutical Co Ltd | Leukemia, Myeloid, Acute | Details |
| AMG-794 | AMG-794 | Phase 1 Clinical | Amgen Inc | Carcinoma, Ovarian Epithelial; Carcinoma, Non-Small-Cell Lung | Details |
| QLS-31904 | QLS31904; QLS-31904 | Phase 1 Clinical | Qilu Pharmaceutical Co Ltd | Solid tumours | Details |
| YK-012 | YK012 | Phase 1 Clinical | Lymphoma, B-Cell | Details | |
| RO-7428731 | RO-7428731; RG-6156 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Glioblastoma | Details |
| GBR-1342 | GBR-1342; ISB-1342 | Phase 1 Clinical | Glenmark Pharmaceuticals Ltd | Multiple Myeloma | Details |
| Recombinant humanized anti-CD19/CD3 bispecific antibody(New Time Pharmaceutical) | LNF-1904 | Phase 1 Clinical | Shandong New Time Pharmaceutical Co Ltd | Lymphoma, B-Cell; Leukemia, B-Cell | Details |
| XmAb-968 | XmAb968 | Phase 1 Clinical | Xencor Inc | Hematologic Neoplasms; Leukemia, Promyelocytic, Acute | Details |
| GR-1803 | GR-1803 | Phase 1 Clinical | Chongqing Zhixiang Jintai Biopharmaceutical Co Ltd, Genrix (Shanghai) Biopharmaceutical Co Ltd | Multiple Myeloma | Details |
| XmAb-18968 | XmAb-18968 | Phase 1 Clinical | The Medical College Of Wisconsin Nonprofit | Leukemia, Myeloid, Acute | Details |
| SQZ-622(Novartis Pharma) | SQZ-622 | Phase 1 Clinical | Novartis Pharma Ag | Leukemia, Myeloid, Acute | Details |
| Forimtamig | RG-6234 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Multiple Myeloma | Details |
| RG-6232 | RG-6232 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Melanoma | Details |
| RG-6007 | RO-7283420; RG-6007 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Leukemia, Myeloid, Acute | Details |
| SAR-443216 | SAR-443216 | Phase 1 Clinical | Sanofi | Solid tumours; Stomach Neoplasms; Neoplasms; Breast Neoplasms; Lung Neoplasms | Details |
| Recombinant oncolytic type II herpes simplex virus (Binhui Biopharmaceutical) | BS-006; oHSV2-BiTEs | Phase 1 Clinical | Wuhan Binhui Biotechnology Co Ltd | Solid tumours; Neoplasms; Melanoma; Uterine Cervical Neoplasms | Details |
| JNJ-78306358 | JNJ-78306358; JNJ-6358 | Phase 1 Clinical | Janssen Research & Development Llc | Solid tumours | Details |
| F-182112 | F-182112; F182112 | Phase 1 Clinical | Shandong New Time Pharmaceutical Co Ltd | Multiple Myeloma | Details |
| TNB-585 | TNB-585; AMG-340 | Phase 1 Clinical | Teneobio Inc | Prostatic Neoplasms, Castration-Resistant | Details |
| BC-3448 | BC3448; BC-3448 | Phase 1 Clinical | Solid tumours | Details | |
| IBI-389 | IBI-389 | Phase 1 Clinical | Innovent Biologics(Suzhou) Co Ltd | Solid tumours; Neoplasms | Details |
| hEGFRvIII-CD3 Bi-scFv | Phase 1 Clinical | Duke University | Glioblastoma; Glioma | Details | |
| JNJ-78278343 | Phase 1 Clinical | Janssen Research & Development Llc | Prostatic Neoplasms, Castration-Resistant; Prostatic Neoplasms | Details | |
| TQB-2825 | TQB-2825 | Phase 1 Clinical | Wuxi Biologics Co Ltd | Hematologic Neoplasms | Details |
| RGV-004 | RGV-004 | Phase 1 Clinical | Hangzhou Rongu Biotechnology Co Ltd | Lymphoma, B-Cell | Details |
| Recombinant humanized anti-BCMA/CD3 bispecific antibody(New Time Pharmaceutical) | Phase 1 Clinical | Shandong New Time Pharmaceutical Co Ltd | Multiple Myeloma | Details | |
| NVG-111 | NVG-111 | Phase 1 Clinical | NovalGen Ltd | Lymphoma, Large B-Cell, Diffuse; Lymphoma, Mantle-Cell; Lymphoma, Follicular; Melanoma; Carcinoma, Non-Small-Cell Lung; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| BI-765049 | BI-765049; OBT-624 | Phase 1 Clinical | Oxford Biotherapeutics Ltd | Solid tumours | Details |
| GNC-039 | GNC-039 | Phase 1 Clinical | Solid tumours; Hematologic Neoplasms; Glioma; Neoplasm Metastasis | Details | |
| RO-7425781 | RO-7425781 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Multiple Myeloma | Details |
| RO-7293583 | RO-7293583 | Phase 1 Clinical | F. Hoffmann-La Roche Ltd | Skin Melanoma; Uveal melanoma; Melanoma | Details |
| Rezetamig | JNJ-8780; JNJ75348780; JNJ-75348780 | Phase 1 Clinical | Teneobio Inc | Lymphoma, B-Cell; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| Y-150(Wuhan Yzy Biopharma/CSPC Pharmaceutical) | Y-150; Y150 | Phase 1 Clinical | Wuhan Yzy Biopharma Co Ltd, CSPC Pharmaceutical Group Ltd | Multiple Myeloma | Details |
| EX-101 | EX-101; EX101 | Phase 1 Clinical | Guangzhou Excelmab Inc | Solid tumours | Details |
| Plamotamab | XmAb-13676 | Phase 1 Clinical | Xencor Inc | Lymphoma, B-Cell; Lymphoma, Large B-Cell, Diffuse; Lymphoma, Non-Hodgkin; Leukemia, Lymphocytic, Chronic, B-Cell | Details |
| A-337 | A-337 | Phase 1 Clinical | Evive Biotech Ltd | Solid tumours; Neoplasms | Details |
| CCW-702 | CCW-702 | Phase 1 Clinical | The Scripps Research Institute Inc, Abbvie Inc | Prostatic Neoplasms | Details |
| Tepoditamab | MCLA-117 | Phase 1 Clinical | Pharmaceutical Research Associates, Institute Gustave-Roussy, Merus Nv, Vu University Medical Center, Lgc | Leukemia, Myeloid, Acute | Details |
| ERY-974 | ERY-974 | Phase 1 Clinical | Chugai Pharmaceutical Co Ltd | Solid tumours; Carcinoma, Hepatocellular | Details |
| InHeAb-01 | InHeAb-01; bsAB | Clinical | University Hospital Tuebingen | Neoplasms | Details |
This web search service is supported by Google Inc.







